Non-Rainfall Moisture & Arid Land Decomposition
Collaborators: Sarah Evans (Michigan State University); Kathy Jacobson (Grinnell College); Peter Jacobson (Grinnell College); Mary Seely (Desert Research Foundation of Namibia); Kathe Todd-Brown (Pacific Northwest National Lab); Anthony Wenndt (Cornell University)
|
For decades, arid ecologists assumed that major processes in deserts are controlled almost exclusively by rainfall. While this is typically the case for primary production, several recent studies have shown that this doesn't hold true for other ecosystem processes like decomposition that are closely tied to sun exposure, spatial heterogeneity, and microbial adaptation. Recent work with our colleagues at Grinnell College has begun to show that non-rainfall moisture sources (fog, dew, and even high humidity) are sufficient to trigger substantial microbial decomposition during inter-rain periods, with large implications for the role of arid lands in global carbon cycling. We are studying how non-rainfall moisture interacts with both microbial communities and abiotic drivers like sunlight and physical weathering to control decomposition in arid and semi-arid lands. This work expands on the photodegradation work by using culture-based assays to measure functional decomposition ability of local microbes in response to different moisture regimes alongside sequencing approaches to quantify community-level changes associated with plant litter.
|
Fog on Stipagrostis sabulicola leaves in the Namib Desert. Many plants and animals in the Namib use fog and dew as water sources and we are starting to understand the role that these non-rainfall moisture inputs influence microbial activity as well. Photo by Robert Logan.
|
Photodegradation in Dry Lands
Collaborators: Sarah Evans (Michigan State University); Kathy Jacobson (Grinnell College); Peter Jacobson (Grinnell College); Paul Barnes (Loyola University New Orleans); Matt Robson (University of Helsinki); Marta Pieristé (University of Helsinki & University of Rouen)
|
The "Very Small Array" of sunlight manipulation shelters at the Gobabeb Research & Training Centre in the Namib Desert. Photo by Oliver Hasley
|
In July 2016, I setup a series of sunlight-manipulation shelters at Gobabeb to study the interaction between sunlight and microbial communities in plant litter decomposition. Gobabeb staff are monitoring twenty four shelters arranged in a grid with four treatments (fully exposed, UV-transparent, UV-blocking, and shade). Over the next three years, I collected samples every six months to track changes in microbial community composition and abundance, litter mass loss, and litter chemistry and I'm currently analyzing the data. Results from this study will eventually help us better model carbon cycling in arid and semi-arid lands.
Here's a slightly longer description of the project that is used for Gobabeb station tours and visiting school groups. |
Drought Resilience of Desert Springs
Collaborators: Peter Jacobson (Grinnell College); Keith Schilling (Iowa DNR); Jeff Muntifering (Save the Rhino Trust, Namibia); Duncan Mitchell (University of the Witwatersrand), David Peate (University of Iowa); Theo Wassenaar (Gobabeb Research and Training Centre)
|
As deserts get drier, reliable water sources, like groundwater springs, are at risk of drying up which can have big effects on wildlife. The most obvious impact is a decrease in water availability as water sources disappear but changes in water quality that come with evaporative concentration can also cause problems for animals. Since 2012, we have been working to map the assess the drought tolerance of groundwater springs in northwestern Namibia. We are using historical and current observations of springs' susceptibility to drought, geochemical and isotopic analysis of groundwater to map aquifers, information about the spatial distribution of springs to identify changes in animal habitat during drought conditions, and behavioral observations of animals in the field to assess their tolerance of changing salinities. This work is being done in partnership with the Save the Rhino Trust of Namibia and is supported by MSU's Africa Studies Center and the Center for Global and Regional Environmental Research at University of Iowa.
|
The Gobabeb Salt Spring has higher salinity than ocean water but several species appear to drink from it. Photo by Reyk Börner.
|
Fog-Dispersed Microbes
Collaborators: Sarah Evans (Michigan State University); Kathie Weathers (Cary Institute of Ecosystem Studies); Eli Dueker (Bard College)
Check out our paper in Science of the Total Environment
Check out our paper in Science of the Total Environment
|
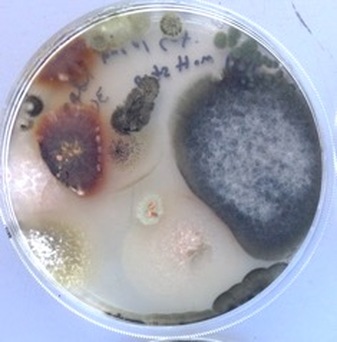
Fungi and bacteria isolated on a malt agar exposed during a fog at Gobabeb and incubated for 5 days. Plates exposed during fog showed higher visual growth relative to controls set out several hours before the fog. Photo by Sarah Evans.
|
Although we have known that atmospheric fog can play a very important role in systems by bringing in biologically available water, we know less about the role fog plays in transporting other resources into systems. These can include chemical inputs like salt and marine-derived nitrogen but we are beginning to see evidence of active microbial communities (including both bacteria and fungi) in fog. In many coastal environments, fog may act as a vector for marine-derived microorganisms, including potential pathogens or other ecologically-relevant microbes, to reach and be deposited on land.
We are working on early characterization of the microbial communities associated with several coastal fog systems to characterize the biodiversity associated with fog and how weather patterns and local conditions drive changes in these communities. |
Groundwater Microbes in Hyperarid Springs
Collaborators: Peter Jacobson (Grinnell College); Jeff Muntifering (Save the Rhino Trust, Namibia); Sarah Evans (Michigan State University); Matt Schrenk (Michigan State University)
|
Although microbes underground make up an enormous proportion of the total biomass and biodiversity of this planet, we still no very little about them. This is mainly because the drilling equipment required to access these subsurface ecosystems is very expensive. For this project, I'm interested in using water emerging from groundwater springs to assess the microbial community of aquifers in Namibia's Kunene region without having to rely on drilling. Linking microbial community and trait data using high-throughput sequencing and trait databases with geological and geochemical data of the water can allow us to ask questions about what drives community assembly in groundwater ecosystems.
This project is done in partnership with the Save the Rhino Trust and is supported through a Graduate Research Fellowship by the Michigan Space Grant Consortium. |
Me sampling groundwater at a highly vegetated spring in western Namibia. Photo by Peter Jacobson.
|